Stefano Artin Serapian and Marc W. van der Kamp
In this recent ACS Catalysis publication, Stefano Serapian and Marc van der Kamp used a variety of computational tools to shed light on an enticing problem in biocatalysis that has proven very difficult to solve experimentally.
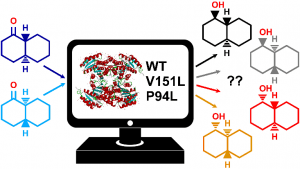
Our enzyme of interest—actinorhodin ketoreductase (actKR)—is found in the soil bacterium Streptomyces coelicolor, where it is normally implicated in the biosynthesis of the antibiotic actinorhodin. In addition to actKR’s natural scope, as is the case with other ketoreductases, some of its re-engineered variants are highly attractive to synthetic chemists by virtue of their high stereoselectivity in reducing small-molecule achiral ketones to chiral alcohols.
Experimental work on actKR featuring the small model substrate trans-1-decalone (a bicyclic aliphatic ketone) and similarly-sized chiral alcohols suggests that whereas the wild-type enzyme is mildly S-selective, some variants (e.g. the P94L mutant) were entirely S- selective, whereas others (e.g. the V151L mutant) only exhibited R- selectivity.
Featuring classical MD, MM/PBSA and hybrid QM/MM MD with umbrella sampling, our study successfully unravels the causes of such remarkable behaviour in wild-type, P94L, and V151L actKR towards trans-1-decalone. Explicitly examining both enantiomers of this naturally racemic substrate (something difficult to achieve in vitro), we conclude that changes in stereocontrol across actKR variants can be dictated by a subtle interplay of different causes (including reaction barrier height and accessibility of reactive poses). Interestingly, however, which factor is dominant in conferring stereoselectivity differs per variant.
The protocols we have used were chosen such that they (1) require input of the WT structure only; (2) use relatively limited computational resources (short simulations and semiempirical QM treatment); and (3) can be automated. Our study is thereby a good example of how computational biochemistry can become a practical, useful and efficient tool in biocatalyst engineering, offering perspectives that might otherwise be difficult to explore in vitro.